|
Basic Concepts in Electrochemistry. 2 Electrochemical Cell Electrons Current + -. Electrolysis Cell Electrons (2 e) Current + - ANODE CATHODE Current Voltage Source. Notes on electrolysis of substances. Books Audiobooks Comics Sheet Music. Hand-picked favorites from our editors. Electro-Chemical reaction Engineering Electrode Reactions. 
Electrolysis - Wikipedia, the free encyclopedia. This article is about the chemical process. 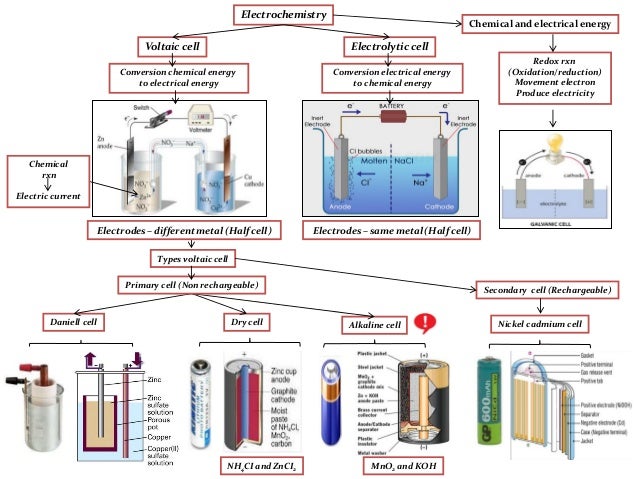
A summary of Electrolysis in 's Electrolytic Cells. Learn exactly what happened in this chapter, scene, or section of Electrolytic Cells and what it means. Chemistry 12 Notes on Unit 5-Redox Electrolytic Cells Chemistry 12—Notes on Electrolytic Cells Page 41 Electrolytic Cells (ELC’s) Electrolysis – uses an. Electrolysis is the break-up of a chemical compound using an electric current. See similar resources. For the cosmetic hair removal procedure, see Electrology. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential. History. Choice of suitable electrode depends on chemical reactivity between the electrode and electrolyte and manufacturing cost. Process of electrolysis. The desired products of electrolysis are often in a different physical state from the electrolyte and can be removed by some physical processes. For example, in the electrolysis of brine to produce hydrogen and chlorine, the products are gaseous. These gaseous products bubble from the electrolyte and are collected. Positively charged ions (cations) move towards the electron- providing (negative) cathode. Negatively charged ions (anions) move towards the electron- extracting (positive) anode. In this process electrons are either absorbed or released. Neutral atoms gain or lose electrons and become charged ions that then pass into the electrolyte. The formation of uncharged atoms from ions is called discharging. When an ion gains or loses enough electrons to become uncharged (neutral) atoms, the newly formed atoms separate from the electrolyte. Positive metal ions like Cu++ deposit onto the cathode in a layer. The terms for this are electroplating, electrowinning, and electrorefining. When an ion gains or loses electrons without becoming neutral, its electronic charge is altered in the process. In chemistry, the loss of electrons is called oxidation, while electron gain is called reduction. Oxidation and reduction at the electrodes. For example, it is possible to oxidize ferrous ions to ferric ions at the anode: Fe. For example: p- Benzoquinone can be reduced to hydroquinone at the cathode: + 2 e. Electrolysis reactions involving H+ ions are fairly common in acidic solutions. In aqueous alkaline solutions, reactions involving OH. It is even possible to have electrolysis involving gases. Such as when using a Gas diffusion electrode. Energy changes during electrolysis. The losses can (in theory) be arbitrarily close to zero, so the maximum thermodynamic efficiency equals the enthalpy change divided by the free energy change of the reaction. In most cases, the electric input is larger than the enthalpy change of the reaction, so some energy is released in the form of heat. In some cases, for instance, in the electrolysis of steam into hydrogen and oxygen at high temperature, the opposite is true and heat energy is absorbed. This heat is absorbed from the surroundings, and the heating value of the produced hydrogen is higher than the electric input. Related techniques. Though related via the interaction of ions and electrodes, electrolysis and the operation of electrochemical cells are quite distinct. A chemical cell should not be thought of as performing electrolysis in reverse. Faraday's laws of electrolysis. This became the basis of the first law of electrolysis: m=k. Electroplating is used in many industries for functional or decorative purposes, as in vehicle bodies and nickel coins. Production of hydrogen for fuel, using a cheap source of electrical energy. Electrolytic etching of metal surfaces like tools or knives with a permanent mark or logo. Electrolysis is also used in the cleaning and preservation of old artifacts. Because the process separates the non- metallic particles from the metallic ones, it is very useful for cleaning a wide variety of metallic objects, from old coins to even larger objects including rustedcast ironcylinder blocks and heads when rebuilding automobile engines. Rust removal from small iron or steel objects by electrolysis can be done in a home workshop using simple materials such as a plastic bucket, tap water, lengths of rebar, washing soda, baling wire, and a battery charger. However, with salts of some metals (e. In both cases this is due to water being reduced to form hydrogen or oxidized to form oxygen. In principle the voltage required to electrolyze a salt solution can be derived from the standard electrode potential for the reactions at the anode and cathode. The standard electrode potential is directly related to the Gibbs free energy, . An extract from the table of standard electrode potentials is shown below. Half- reaction. E. For example, it is more difficult to reduce a sodium ion to a sodium metal than it is to reduce a zinc ion to a zinc metal. Reduced species (often an anion) with a more positive cell potential are more difficult to oxidize than reduced species with a more negative cell potential. For example, it is more difficult to oxidize sulfate anions than it is to oxidize bromide anions. Using the Nernst equation the electrode potential can be calculated for a specific concentration of ions, temperature and the number of electrons involved. For pure water (p. H 7): the electrode potential for the reduction producing hydrogen is . The conclusion from these figures is that hydrogen should be produced at the cathode and oxygen at the anode from the electrolysis of water. In practice many other factors have to be taken into account such as the kinetics of some of the reaction steps involved. These factors together mean that a higher potential is required for the reduction and oxidation of water than predicted, and these are termed overpotentials. Experimentally it is known that overpotentials depend on the design of the cell and the nature of the electrodes. For the electrolysis of a neutral (p. H 7) sodium chloride solution, the reduction of sodium ion is thermodynamically very difficult and water is reduced evolving hydrogen leaving hydroxide ions in solution. At the anode the oxidation of chlorine is observed rather than the oxidation of water since the overpotential for the oxidation of chloride to chlorine is lower than the overpotential for the oxidation of water to oxygen. The hydroxide ions and dissolved chlorine gas react further to form hypochlorous acid. The aqueous solutions resulting from this process is called electrolyzed water and is used as a disinfectant and cleaning agent. Research trends. The efficiency of an electrolyser is a measure of the enthalpy contained in the hydrogen (to undergo combustion with oxygen, or some other later reaction), compared with the input electrical energy. Heat/enthalpy values for hydrogen are well published in science and engineering texts, as 1. MJ/kg. Note that fuel cells (not electrolysers) cannot utilise this full amount of heat/enthalpy, which has led to some confusion when calculating efficiency values for both types of technology. In the reaction, some energy is lost as heat. Some reports quote efficiencies between 5. PEM (Polymer Electrolyte Membrane electrolysis) and catalytic technology, such as 9. Hydrogen is used for the creation of ammonia for fertilizer via the Haber process, and converting heavy petroleum sources to lighter fractions via hydrocracking. Electrocrystallization. The technique has been used to obtain single crystals of low- dimensional electrical conductors, such as charge- transfer salts. The Chemical news and journal of industrial science; with which is incorporated the . Retrieved 2. 7 February 2. Understanding solids: the science of materials. ISBN 9. 78- 0- 4. Retrieved 2. 2 October 2. Retrieved April 1, 2. Physical Chemistry, 6th edition (W. H. Freeman and Company, New York).^ abc. Van. Vogel, 1. 95. A textbook of Quantitative Inorganic Analysis, Longmans, Green and Co^Y. Hori, in Modern Aspects of Electrochemisty , ed. Gamboa- Aldeco, Springer, New York, 2. Qiao, et al., A review of catalysts for the electroreduction of carbon dioxide to produce low- carbon fuels, Chem. Soc. Rev., 2. 01. Carmo, M; Fritz D; Mergel J; Stolten D (2. Journal of Hydrogen Energy. Part 4: Production from electricity by means of electrolysis. Sverdrup (March 2. National Renewable Energy Laboratory. Retrieved 2. 0 October 2. Synthesis, structure, and conductivity of bis(tetramethyltetraselenafulvalenium) perchlorate, (TMTSF)2. Cl. O4. Journal of the American Chemical Society. IB Chemistry higher level notes: Electrolysis. Solutions in water contain hydrogen. These H+ and. OH- ions compete at the electrodes with the solute ions. The ions that are successfully released at the electrodes depend on three. The position of the ion in the electrochemical series. The concentration of the ion in the solution. The nature of the electrode. The position of the ion in the electrochemical series. This is probably better expressed as the position of the redox equilibrium. All the redox equilibria are expressed. Cu. 2+ + 2e. Cu However, going from the right hand side to the left hand. Hence it may be called a redox equilibrium. This is more positive than hydrogens electrode potential of. V. This means that copper ions are more easily reduced (i. Consequently in a competition between the two ions the copper. As a rule of thumb, if the metal appears below hydrogen. The concentration of the ions. When two ions with similar reactivity are in competition. The nature of the electrode. Usually, inert electrodes such as graphite or platinum are used for electrolysis. However, if metal electrodes are used in metal ion solutions they can. Example: Electrolysis of sodium chloride. The ions present in the solution are. Na+. . Cl- . . H+. OH- . At the cathode. The positive ions are attracted to the negative cathode. There. is competition between the sodium ions and the hydrogen ions. As. the hydrogen ion . The chloride ions compete with the hydroxide. Whe the solution is. Cl- - 2e. Cl. 2Ions remaining in solution. The ions that are removed from the solution, then. This means that the. Note: When the solution of chloride ions is dilute. OH. There. is competition between the copper ions and the hydrogen ions. As. the hydrogen ion . The sulphate ions compete with the hydroxide. The hydroxide ions. AS OXYGEN GAS and water. OH- - 4e. 2. H2. O + O2. Ions remaining in solution. The ions that are removed from the solution, then. This means that the. Example: Electrolysis of copper(II) sulphate. The ions present in the solution are. Cu. 2+. SO4. 2- . H+. . OH- . At the cathode. The positive ions are attracted to the negative cathode. There. is competition between the copper ions and the hydrogen ions. As. the hydrogen ion . Cu - 2e. Cu. 2+Ions remaining in solution. Copper is deposited at the cathode and is dissolved. Consequently the concentration of copper ions in solution. This can be used as a method of purification of. In this purification an anode made of impure copper. Summary. Reactive metals (more reactive than hydrogen) are never deposited during. If the metal ion comes from a metal. Halide ions (chloride, bromide, iodide) are released preferentially and. Unreactive metals, such as copper or nickel, may participate in reactions. List the factors affecting the amount of product. Factors are charge on the ion, current and. Products of electrolysis. The ions involved in elecrolysis are picking up electrons and depositing. It is apparent that the number of ions involved. It will also depend on the charges of the ions involved. Relationship between current and number of electrons. Each electron posseses an electrical charge and the movement of the electrons. Q'. This is related to the electrical current as follows: 1 Ampere of current (I) is the passage of 1 Coulomb (C) of charge per. To calculate the number of coulombs of charge it is necessary to multiply. One mole of electrons has a charge equivalent to 9. Faraday of charge (F)Charge on the ion. So, to release one mole of a singly charged ion at an electrode exactly. Faraday of charge (9. C) must pass through the electrode. If the ion has a double charge then two moles of electrons (2 Faradays). Example Calculate the number of moles of hydrogen. Then: -Q = 5 x 3. C Therefore: -Q = 1. Faradays In the electrolysis the hydrogen gas is released at the cathode. H+ + 2e. H2. 2 moles of electrons release 1 mole of gas. Faradays of charge are needed to release 1 mole of hydrogen. Therefore 0. 1. 55 F releases 0. Therefore moles of hydrogen released = 0. Example Calculate the number of coulombs needed to. No of moles Cu =. In electrolysis copper metal is released at the cathode as follows: Cu. Cu 2 moles of electrons release 1 mole of copper. Therefore 0. 2 moles of electrons release 0. Faradays = 0. 2 x 9. Calculate the time that a current of 4 amps. Q = It. 19,3. 00 coulombs = It. Therefore t = 1. 9,3. Determine the relative amount of products formed. Relative quantities of product. As ions have different charges it follows that diffierent ions will require. For example, copper ions have a 2+ charge, whereas silver ions have a. It follows, then, that twice as many electrons are. Therefore for the same number of moles of electrons - i. Example. In the following diagram the number of moles of silver deposited will. The number of moles. Useful links. Electrolysis. Purdue Universityhttp: //members. Electrolysis. Industrial.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2017
Categories |
 RSS Feed
RSS Feed
